This article was published in February 2019 and updated in November 2021.
Table of Contents
A couple of years ago, my psych nurse suggested we send some of my cheek cells to a lab for genetic testing. I was severely depressed and hadn’t had much success with the antidepressants I’d tried. With a significant family history of depression and other mental health issues, it seemed likely that there was a genetic component for me.
L-methylfolate: my silver bullet?
My psych nurse told me that for some people, a genetic abnormality affects the activity of a particular enzyme that’s implicated in psychiatric conditions (and many other illnesses). People with this mutation can be helped by taking what’s essentially a supplement (I believe she even said it was “like a vitamin”), l-methylfolate. By taking the product of this hindered metabolic process in pill form, it fills in the gaps that your faulty enzymes have left behind. Because it was a rather serious situation, we decided that we may as well add this supplement to see if it helped. I told her I’d think about the genetic testing.
A couple of weeks later, we revisited the idea. The l-methylfolate seemed to be helping a little, but not dramatically. I had figured that the testing would be unnecessary; if the l-methylfolate helped, we’d know I was a mutant. If it didn’t, we’d move on to something else. It wasn’t that cut and dry.
Why the reluctance, you ask?
Perhaps there was a little anxiety about what I might find out about myself, but I suppose that’s a topic for my therapist. Mostly, I was skeptical. Genetic research has advanced tremendously, but the mechanistic aspects of mental illnesses are still rather poorly understood. Could a DNA profile really tell me why I was depressed?
Well, I’ll spoil the ending for you and say “Yes. Kind of.” Curiosity won out, and I agreed to part with some of my genetic material in the name of science and not wanting to be depressed forever. The results, pared down to what’s important for clinicians and patients to understand, were interesting- and rather ambiguous.
The science behind MTHFR mutations
The gene in question is called methylenetetrahydrofolate reductase (MTHFR). Our genes code for proteins, and enzymes like MTHFR are one type of protein. MTHFR is involved in something called one-carbon metabolism, which has effects on the production of neurotransmitters.
First, some genetics background
Harken back to your high school or college biology class; remember alleles? A gene and an allele are not exactly the same thing. A gene is essentially a small segment of a chromosome. The DNA sequence of a gene codes for a specific sequence of amino acids, which comprise proteins. An allele, however, is a variation of a gene. For each gene, you got one allele from your mom and one from your dad. I received a combination of normal and mutated MTHFR genes. Mutation is how variation occurs, and the process itself is neither good nor bad; it just is. Some mutations are adaptive, some are neutral, and some are deleterious.
Two MTHFR mutations
There are two main mutations on the same allele that seem to affect this enzyme’s activity. As usual, the names are cryptic; they’re called C677T and A1298C. The numbers and letters refer to where in the allele one nucleotide (or one “digit” in the DNA code) has been changed to another, and the substitution that’s taken place. In general genetics contexts, heterozygotes have different alleles from mom and dad, while homozygotes have identical alleles.
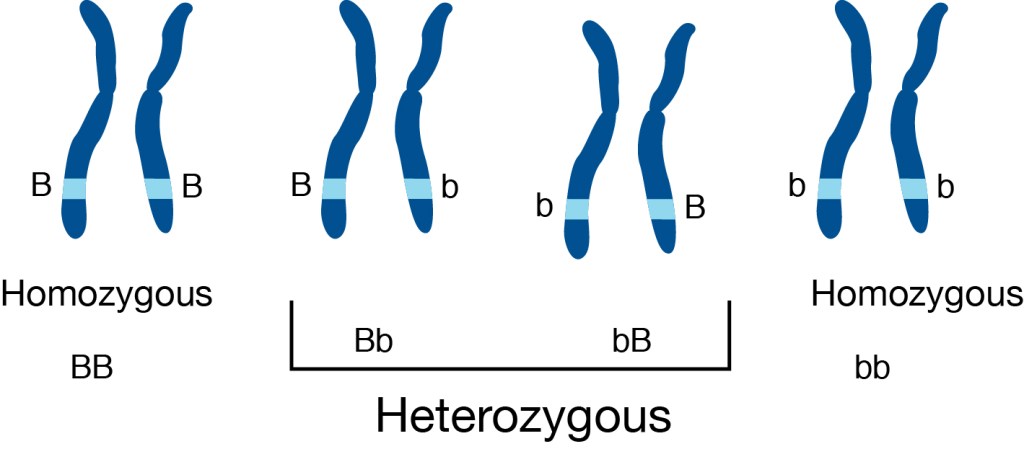
In the context of mutation research, there is typically one genotype that is referred to as “wild type,” meaning it contains no mutations and has full function. In this case, imagine the big “B” homozygous chromosome in the image above as the “wild type” and the little “b” as the mutation in question. The letters in examples like this are simply ways to represent alleles in a theoretical framework. The order of the letters is written out because, without further research, it’s difficult to tell which parent the mutation came from.
To bring it around to MTHFR, I’m a heterozygote for both of the main mutations (this is called “compound heterozygosity”). My genotype for C677T is C/T. The C (cytosine) is the nucleotide in the normal allele and the T (thymine) is the substituted nucleotide in my mutated version. I got the C from one parent and the T from the other. I’m also a heterozygote when it comes to the A1298C mutation. My genotype is A/C, where the A (adenosine) is the normal one and the C is the mutant.
The problem lies in the metabolism of a key metabolic precursor: a biologically-active form of folate. The product of this reaction is 5-tetrahydrofolate, which provides a methyl group for epigenetic regulation. In other words, when MTHFR is mutated, the system that controls gene expression and, ultimately, neurotransmitter production, is affected by a lack of this enzyme’s product because the enzyme’s function is reduced. This is the gap that the supplement I take fills in.
Enzyme activity
Meta-analyses suggest that C677T homozygous mutants (they have two mutated copies) have a 75% reduction in MTHFR activity, and A1298C homozygous mutants have a 39% reduction in enzyme activity. Heterozygotes for each mutation have less severe reductions in activity, but if you’re a compound heterozygote (like me), that results in a 52% reduction. So, I have slightly less than half the enzymatic activity for MTHFR than a normal, non-mutant.
MTHFR and depression
That all seems pretty straightforward, but take a look at the plethora of studies that exist on the internet, and you’ll see why I was cautious. Some have found significant relationships between MTHFR polymorphisms and psychiatric conditions, and yet others haven’t. There are some studies that say that there is no difference between mutant and control subjects when it comes to depression. Others suggest that carrying a mutated MTHFR allele predicts depression when the person is exposed to childhood trauma.
Most of the studies I’ve seen, including meta-analyses, lean towards accepting a link between MTHFR and depression, especially for homozygotes, who have the least MTHFR activity. Experiments have shown evidence that active folate supplementation can be an effective adjunct therapy for treatment-resistant depression.
The consensus seems to be that more research is needed, although increasingly, there does seem to be a relationship between MTHFR and depression.
For me, I’m content to believe that at least some of my struggles can be traced back to MTHFR. The supplement does seem to make my antidepressants more effective. Plus, that’s not all that my genetic report showed. I also have a weird serotonin transporter that makes SSRIs less effective and increases my cortisol release in response to stress. I’m glad I did the test, at the very least because it helped me to accept my disorder as valid and offered insights into my mental health..
The future of mental health treatment
Is this a sure-fire way to treat mental illness? I’d say not yet, but it’s certainly valuable. Personalized medicine seems to be a buzzword floating around these days, and the implications of genetic research for how we approach mental illness are fascinating. Maybe when the relationships between genes and mental illness are elucidated a little more clearly, we can finally kick the stigma and misconceptions out the door. One can hope, at least.
(Featured image credited to: Abogomazova – Own work, CC BY-SA 3.0, https://commons.wikimedia.org/w/index.php?curid=25074990)